60601-1 edition 3.1 and understanding IEC60601-1 Document structure
Globtek applies for IEC 60601-1 medical Safety Approvals and EMC approvals on their standard power supply products. The medical approvals for Safety and EMC are required to exist in separate CB reports.
Many medical products are used in a home health care environment. Globtek obtains a IEC 60601-1-11 approval on their products in order to support this important market segment of the medical equipment business. An important distinction with home use medical equipment is that the AC input configuration must be a Class II (unearthed, 2 conductor) configuration.
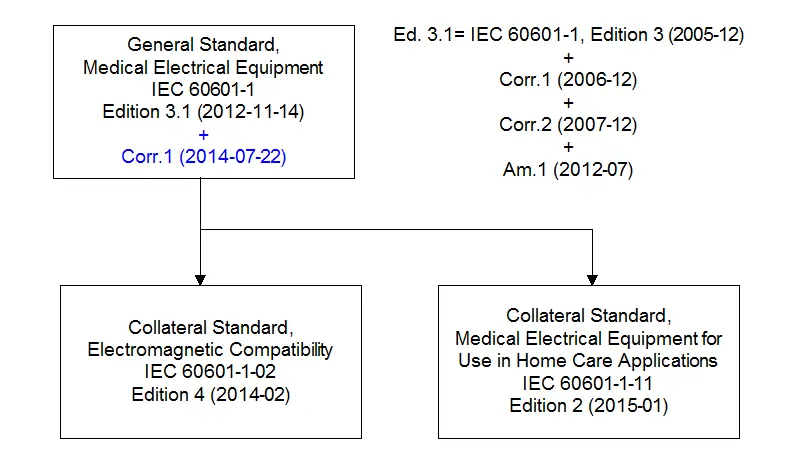
There is often confusion regarding the differences between IEC 60601-1 ed 3, and IEC 60601-1 ed 3.1. As shown in the diagram below, when Corrigendum 1 and 2, and Amendment 1 are applied to edition 3, the resulting document is the same as edition 3.1. The changes are generally minor between ed 3 and ed 3.1, one major change is to have relaxed safety isolation barrier requirements for low voltage DC systems, such as automobile batteriesl, where the risk is far less than a 230Vac input safety barrier.
In the diagram below, the two key collateral standards for IEC 60601-1 which Globtek obtains CB approvals (and specific harmonized national standards approvals) for are shown, which is for the medical EMC standard and the Home Use Care standard.
For additional information on this topic, please visit the following web pages:
- MECA (Medical Equipment Compliance Associates) US Home Page: http://mecassociates.us/
- MECA, list of 60601-1 standards, collateral standards, and particular standards: http://60601-1.com/assets/meca-60601-80601-medical-standards-list-(rev-2015-12-24).pdf
GlobTek Products which meet IEC60601 3.1
| Model | Type | Input Voltage | Watts | Vout | Efficiency | Ingress Protection | MOOP | MOPP | 60601-1-4th Ed. | IEC 62368-1 | Dimensions (mm) |
|---|---|---|---|---|---|---|---|---|---|---|---|
| GTM96060-06VV-Q | Wall Plug-in | 100-240V~, 50-60 Hz | 6 | 3.3-48 V | USA DOE Level VI / Eco-design Directive 2009/125/EC, (EU) 2019/1782 | IP42 (Optional IP54-IP55) | ✔ | ✔ | ✔ | 43.5*74.0*36.8 | |
| GTM41134-WWVV-F | Open Frame/Internal | 100-240V~, 50-60 Hz | 6 | 3.3-48 V | N/A | ✔ | ✔ | ✔ | 84.5*41.5*17.0 | ||
| GTM46101-WWVV-X.X-USB | Wall Plug-in | 100-240V~, 50-60 Hz | 10 | 5-5.5 V | USA DOE Level VI / Eco-design Directive 2009/125/EC, (EU) 2019/1782 | Indoor Use | ✔ | ✔ | ✔ | 41.0 x 71.0 x 31.5 | |
| GTM43007-CWWVV-F | Open Frame/Internal | 100-240V~, 50-60 Hz | 60 | 5-48 V | N/A | N/A | ✔ | ✔ | ✔ | 127x76.2x23.6 | |
| GTM91099-60VV-T2 | Wall Plug-in+Desktop Combination | 100-240V~, 50-60 Hz | 60 | 5-48 V | V | IP42 | ✔ | ✔ | 117.53*53.47*36.6 | ||
| GTM43007-CWWVV-FW | Open Frame/Internal | 100-240V~, 50-60 Hz | 60 | 5-48 V | N/A | N/A | ✔ | ✔ | ✔ | 127x76.2x23.6 | |
| GTM43007-BWWVV-F | Open Frame/Internal | 100-240V~, 50-60 Hz | 60 | 5-48 V | N/A | N/A | ✔ | ✔ | ✔ | 101.6x50.8x23.6 for 5V-14V models, 101.6x50.8x22.5 for 15V-48V models | |
| GTM43007-AWWVV-FW | Open Frame/Internal | 100-240V~, 50-60 Hz | 60 | 5-48 V | N/A | N/A | ✔ | ✔ | ✔ | 76.2x50.8x23.6 for 5V-14V models, 76.2x50.8x22.5 for 15V-48V models | |
| GTM43007-BWWVV-FW | Open Frame/Internal | 100-240V~, 50-60Hz | 60 | 5-48 V | N/A | N/A | ✔ | ✔ | ✔ | 101.6x50.8x23.6 for 5V-14V models, 101.6x50.8x22.5 for 15V-48V models | |
| GTM91099-60VV-T3A | Wall Plug-in+Desktop Combination | 100-240V~, 50-60 Hz | 60 | 5-48 V | V | IP52, IP54 Option available | ✔ | ✔ | ✔ | 117.53*53.47*36.6 | |
| GTM43007-AWWVV-F | Open Frame/Internal | 100-240V~, 50-60 Hz | 60 | 5-48 V | N/A | N/A | ✔ | ✔ | ✔ | 76.2x50.8x23.6 for 5V-14V models, 76.2x50.8x22.5 for 15V-48V modelsTop Diagram i |