IEC and CENELEC Standard Naming Conventions: 60601-1-2 4th Edition vs. EN 60601-1-2:2015 vs IEC 60601-1-2:2014-02
The International Electrotechnical Commission (IEC) is the leading global organization that prepares and publishes International Standards for all electrical, electronic and related technologies. Both Safety and Electromagnetic compliance standards are produced by the IEC.
Virtually all countries in the world rely on and adopt the IEC standards, in part to simplify creating their own standards, and in part to harmonize their country standards with the world. However most countries choose to not directly use the IEC standards, but instead create derivative standards directly based on the IEC standards. In this way it is possible to add minor changes to the adopted standards on an as needed basis.
CENELEC is the European agency which typically controls the EN standards. Most of the EN electro-technical standards produced by CENELEC are directly derived from IEC standards, and typically the major numeric part of standard name is identical. Differences in the standard number however may occur in the suffix which will typically indicate year of publication as 4 digits. CENELEC and standards published by various countries typically will lag the publication of the IEC standard by one or more years.
Additionally, whereas Edition levels are used for many of the IEC standards to clarify to the user a major change to the standard, this is not practiced in the CENELEC naming convention of EN standards.
The IEC recommends a 5 year allowance for manufacturers to modify their products before the derived national standards, become mandatory, and any conflicting national standards are withdrawn. Therefore when countries adopt a new version/edition of an IEC standard, two key dates are typically specified in the new derived standard:
- DOP, Date of Publication
- DOW, Date of Withdrawal
These two key dates are typically shown within the first 5 pages of an EN specification, and the CENELEC requirements for DOW mostly follow the IEC recommendations.
As an example, let us consider the medical EMC control document IEC 60601-1-2-2014, 4th edition.
The full numeric descriptor of the standard is IEC 60601-1-2:2014-02. The year and month of publication follow the colon which is attached to the primary specification number. Although the 4th edition does not appear in the number, reviewing the Title page of the standard shows it prominently displayed. After this IEC edition was published, subsequently in January 2015, the CENELEC Standard EN 60601-1-2:2015 was ratified. Note, only the year is shown as part of the standard name, the month is not included.
The DOP for the EN 60601-1-2:2015 is 2016-03-18, the DOW is 2018-12-31. After 2018-12-31 all products sold in the EU are required to meet the requirements in this medical EMC standard.
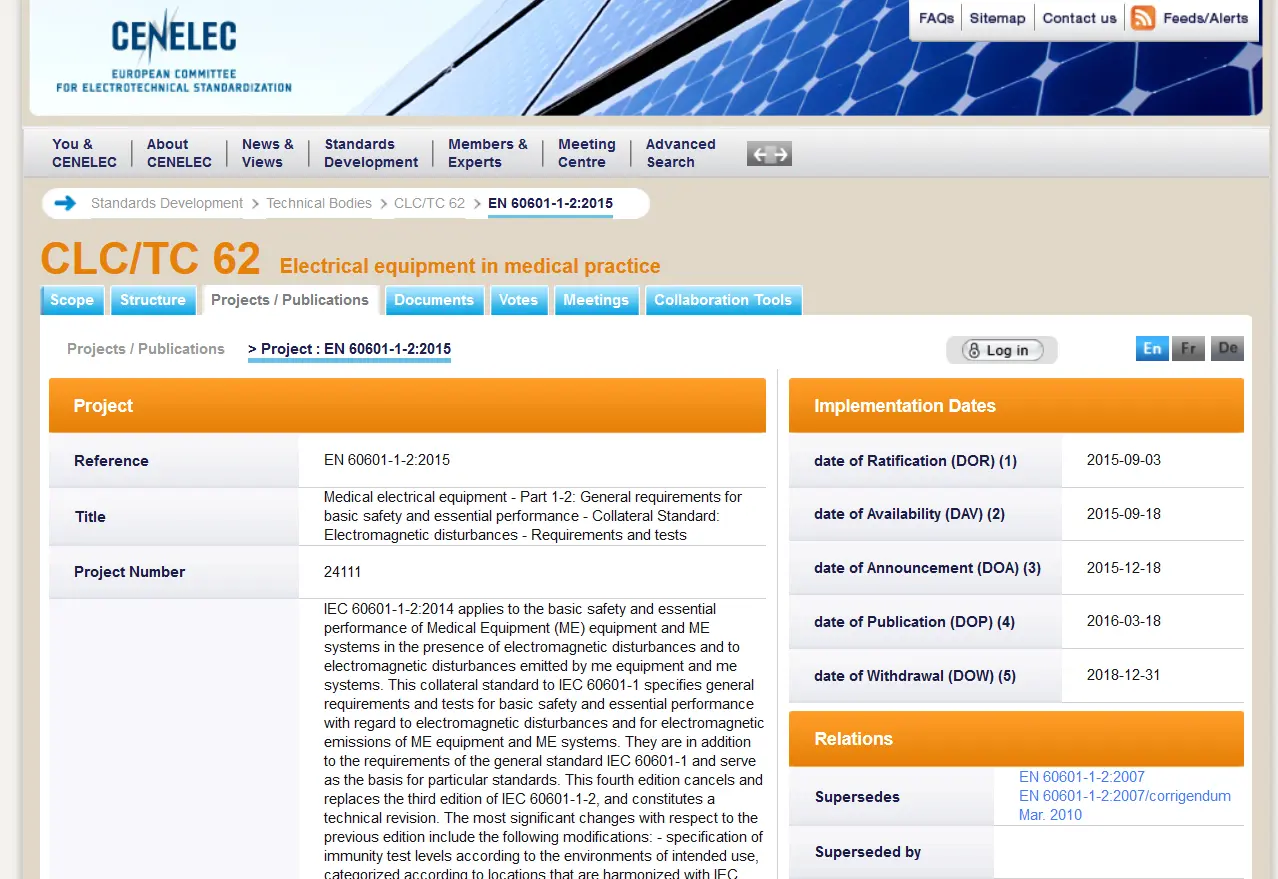
REF: https://www.cenelec.eu/dyn/www/f?p=104:110:974359894211801::::FSP_ORG_ID,FSP_PROJECT,FSP_LANG_ID:1257161,44445,25
GlobTek products compliant with IEC 60601-1-2 4th edition:
| Name | Type | Input Voltage | Watts | Vout | Efficiency | Ingress Protection | MOOP | MOPP | 60601-1-4th Ed. | IEC 62368-1 | Dimensions (mm) |
|---|---|---|---|---|---|---|---|---|---|---|---|
| GT-41134-W2-TAB | Wall Plug-in | 120V~/60Hz | 6 | 6 V | V | IP40 | ✔ | ✔ | |||
| GTM41134-WWVV-F | Open Frame/Internal | 100-240V~, 50-60 Hz | 6 | 3.3-48 V | N/A | ✔ | ✔ | ✔ | 84.5*41.5*17.0 | ||
| GTM41134-WWVV-FWT2 | Open Frame/Internal | 100-240V~, 50-60 Hz | 6 | 5 V | N/A | ✔ | ✔ | ✔ | 84.5*41.5*17.0 | ||
| GTM96060-06VV-Q | Wall Plug-in | 100-240V~, 50-60 Hz | 6 | 3.3-48 V | USA DOE Level VI / Eco-design Directive 2009/125/EC, (EU) 2019/1782 | IP42 (Optional IP54-IP55) | ✔ | ✔ | ✔ | 43.5*74.0*36.8 | |
| GTM46101-WWVV-X.X-USB | Wall Plug-in | 100-240V~, 50-60 Hz | 10 | 5-5.5 V | USA DOE Level VI / Eco-design Directive 2009/125/EC, (EU) 2019/1782 | Indoor Use | ✔ | ✔ | ✔ | 41.0 x 71.0 x 31.5 | |
| GTM46161-16V.V-USB | Wall Plug-in | 100-240V~, 50-60 Hz | 16 | 5 V | USA DOE Level VI / Eco-design Directive 2009/125/EC, (EU) 2019/1782 | Indoor Use | ✔ | ✔ | ✔ | 74.80*41.00*35.30 | |
| GTM46161-16V.V-USB1A | Wall Plug-in | 100-240V~, 50-60 Hz | 16 | 5-5.5 V | USA DOE Level VI / Eco-design Directive 2009/125/EC, (EU) 2019/1782 | Indoor Use | ✔ | ✔ | ✔ | 41.0 x 71.0 x 31.5 | |
| GTM46161-16V.V-USB1A1C | Wall Plug-in | 100-240V~, 50-60 Hz | 16 | 5-5.5 V | USA DOE Level VI / Eco-design Directive 2009/125/EC, (EU) 2019/1782 | Indoor Use | ✔ | ✔ | ✔ | 41.0 x 70.0 x 35.1 | |
| GTM46161-16V.V-USB2A | Wall Plug-in | 100-240V~, 50-60 Hz | 16 | 5-5.5 V | USA DOE Level VI / Eco-design Directive 2009/125/EC, (EU) 2019/1782 | Indoor Use | ✔ | ✔ | ✔ | 41.0 x 70.0 x 35.1 | |
| GTM46161-16V.V-USB2C | Wall Plug-in | 100-240V~, 50-60 Hz | 16 | 5-5.5 V | USA DOE Level VI / Eco-design Directive 2009/125/EC, (EU) 2019/1782 | Indoor Use | ✔ | ✔ | ✔ | 41.0 x 70.0 x 35.1 | |
| GTM46161-16V.V-USBC | Wall Plug-in | 100-240V~, 50-60 Hz | 16 | 5-5.5 V | USA DOE Level VI / Eco-design Directive 2009/125/EC, (EU) 2019/1782 | Indoor Use | ✔ | ✔ | ✔ | 41.0 x 70.0 x 35.1 | |
| GTM41080-18VV-F | Open Frame/Internal | 100-240V~, 50-60 Hz | 18 | 5-48 V | V | IPX0 | ✔ | ✔ | ✔ | 74*44*34 | |
| GTM41080-18VV-F | Open Frame/Internal | 100-240V~, 50-60 Hz | 18 | 5-48 V | V | IPX0 | ✔ | ✔ | ✔ | 84.5*41.5*21.6 | |
| GTM96180-18VV H/M | Wall Plug-in | 100-240V~, 50-60 Hz | 18 | 3-48 V | USA DOE Level VI / Eco-design Directive 2009/125/EC, (EU) 2019/1782 | IP42 (Optional IP54-IP55) | ✔ | ✔ | ✔ | 74*43.5*36.8 | |
| GTM96180-18VV-T2 | Desktop/External | 100-240V~, 50-60 Hz | 18 | 5-48 V | USA DOE Level VI / Eco-design Directive 2009/125/EC, (EU) 2019/1782 | IP42 | ✔ | ✔ | ✔ | (L)87x(W)47x(H)32mm | |
| GTM96180-18VV-T2A | Desktop/External | 100-240V~, 50-60 Hz | 18 | 5-48 V | USA DOE Level VI / Eco-design Directive 2009/125/EC, (EU) 2019/1782 | IP42 | ✔ | ✔ | ✔ | (L)87x(W)47x(H)32mm | |
| GTM96180-18VV-T3 | Desktop/External | 100-240V~, 50-60 Hz | 18 | 5-48 V | USA DOE Level VI / Eco-design Directive 2009/125/EC, (EU) 2019/1782 | IP42 | ✔ | ✔ | ✔ | (L)87x(W)47x(H)32mm | |
| GTM96180-18VV-T3A | Desktop/External | 100-240V~, 50-60 Hz | 18 | 5-48 V | USA DOE Level VI / Eco-design Directive 2009/125/EC, (EU) 2019/1782 | IP42 | ✔ | ✔ | ✔ | (L)87x(W)47x(H)32mm | |
| GTM41060-25VV-FW-IM | Open Frame/Internal | 100-240V~, 50-60 Hz | 25 | 3-48 V | Meets/Exceeds for Medical | ✔ | ✔ | 98*50*28 | |||
| GTM91120-30VV-F | Open Frame/Internal | 100-240V~, 50-60 Hz | 30 | 5-48 V | V | ✔ | ✔ | ✔ | ✔ | 101.6x50.8x22.30 | |
| GTM91120-30VV-F (PCB Mounted) | Open Frame/Internal | 100-240V~, 50-60 Hz | 30 | 12-24 V | V | ✔ | ✔ | ✔ | ✔ | 101.6x50.8x22.30 | |
| GTM91120-30VV-FW | Open Frame/Internal | 100-240V~, 50-60 Hz | 30 | 5-48 V | V | ✔ | ✔ | ✔ | ✔ | 101.6x50.8x22.30 | |
| GTM91120-30VV-FW (PCB Mounted) | Open Frame/Internal | 100-240V~, 50-60 Hz | 30 | 5-48 V | V | ✔ | ✔ | ✔ | ✔ | 101.6x50.8x22.30 | |
| GTM91120-30VV-P2 | Potted/Encapsulated in plastic housing | 100-240V~, 50-60 Hz | 30 | 5-48 V | V | ✔ | ✔ | ✔ | 102*46*37 | ||
| GTM91120-30VV-P2(SR with EU Plug 2 pin) | Potted/Encapsulated in plastic housing | 100-240V~, 50-60 Hz | 30 | 5-9 V | V | IP68 | ✔ | ✔ | ✔ | ✔ | |
| GTM91120-30VV-P2(SR with NA Plug 2 pin) | Potted/Encapsulated in plastic housing | 100-240V~, 50-60 Hz | 30 | 5-48 V | V | IP68 | ✔ | ✔ | ✔ | ✔ | |
| GTM91120-30VV-P2(SR with UK Plug 2 pin) | Potted/Encapsulated in plastic housing | 100-240V~, 50-60 Hz | 30 | 5 V | V | IP68 | ✔ | ✔ | ✔ | ✔ | |
| GTM91120-30VV-P3 | Potted/Encapsulated in plastic housing | 100-240V~, 50-60 Hz | 30 | 5-48 V | V | IP68 | ✔ | ✔ | ✔ | 102*46*37 | |
| GTM91120-30VV-P3 (SR with EU Plug 3 pin) | Potted/Encapsulated in plastic housing | 100-240V~, 50-60 Hz | 30 | 5-48 V | V | IP68 | ✔ | ✔ | ✔ | ||
| GTM91120-30VV-P3 (SR with NA Plug 3 pin) | Potted/Encapsulated in plastic housing | 100-240V~, 50-60 Hz | 30 | 5-48 V | V | IP68 | ✔ | ✔ | ✔ | ||
| GTM91120-30VV-T2 | Wall Plug-in+Desktop Combination | 100-240V~, 50-60 Hz | 30 | 5-48 V | V | IP40 (cord), IP52(R-blades) | ✔ | ✔ | ✔ | (L)101.77x(W)47.47x(H)36.7 | |
| GTM91120-30VV-T3A | Wall Plug-in+Desktop Combination | 100-240V~, 50-60 Hz | 30 | 5-48 V | V | IP40 (cord), IP52(R-blades) | ✔ | ✔ | ✔ | ✔ | (L)102.18x(W)47.50x(H)36.7 mm |
| GTM96300-3656-T3-AP | Desktop/External | 100-240V~, 50-60 Hz | 36 | 56 V | USA DOE Level VI / Eco-design Directive 2009/125/EC, (EU) 2019/1782 | IP40 | ✔ | ✔ | ✔ | 107*55*36 | |
| GTM96300-36VV-P2 | Desktop/External | 100-240V~, 50-60 Hz | 36 | 5-9.1 V | USA DOE Level VI / Eco-design Directive 2009/125/EC, (EU) 2019/1782 | IP68 | ✔ | ✔ | ✔ | 102*46*37 | |
| GTM96300-36VV-P3 | Desktop/External | 100-240V~, 50-60 Hz | 36 | 0 V | USA DOE Level VI / Eco-design Directive 2009/125/EC, (EU) 2019/1782 | IP68 | ✔ | ✔ | ✔ | 102*46*37 | |
| GTM96300-36VV-R2 | Wall Plug-in+Desktop Combination | 100-240V~, 50-60 Hz | 36 | 5-54 V | USA DOE Level VI / Eco-design Directive 2009/125/EC, (EU) 2019/1782 | IP40 (cord), IP52(R-blades) | ✔ | ✔ | ✔ | (L)101.73x(W)46.47x(H)38.2mm | |
| GTM96300-36VV-R3A | Wall Plug-in+Desktop Combination | 100-240V~, 50-60 Hz | 36 | 5-54 V | USA DOE Level VI / Eco-design Directive 2009/125/EC, (EU) 2019/1782 | IP40 (cord), IP52(R-blades) | ✔ | ✔ | ✔ | (L)101.7x(W)46.47x(H)38.2mm | |
| GTM96300-36VV-T2 | Desktop/External | 100-240V~, 50-60 Hz | 36 | 5-54 V | USA DOE Level VI / Eco-design Directive 2009/125/EC, (EU) 2019/1782 | IP42, IP54 Option available | ✔ | ✔ | ✔ | (L)107.2.0x(W)50.0x(H)35.50mm | |
| GTM96300-36VV-T2A | Desktop/External | 100-240V~, 50-60 Hz | 36 | 5-54 V | USA DOE Level VI / Eco-design Directive 2009/125/EC, (EU) 2019/1782 | IP42, IP54 Option available | ✔ | ✔ | ✔ | (L)107.2.0x(W)50.0x(H)35.50mm | |
| GTM96300-36VV-T3 | Desktop/External | 100-240V~, 50-60 Hz | 36 | 5-54 V | USA DOE Level VI / Eco-design Directive 2009/125/EC, (EU) 2019/1782 | IP42, IP54 Option available | ✔ | ✔ | ✔ | 107.2x(W)50.0x(H)35.0mm | |
| GTM46402-40VV-x.x-Q | Wall Plug-in | 100-240V~, 50-60 Hz | 40 | 5-48 V | USA DOE Level VI / Eco-design Directive 2009/125/EC, (EU) 2019/1782 | Indoor Use | ✔ | ✔ | ✔ | 43.5 x 74.0 x 35.3 | |
| GTM43007-AWWVV-F | Open Frame/Internal | 100-240V~, 50-60 Hz | 60 | 5-48 V | N/A | N/A | ✔ | ✔ | ✔ | 76.2x50.8x23.6 for 5V-14V models, 76.2x50.8x22.5 for 15V-48V modelsTop Diagram i | |
| GTM43007-AWWVV-F (D3 with standoffs) | Open Frame/Internal | 100-240V~, 50-60 Hz | 60 | 5-48 V | N/A | N/A | ✔ | ✔ | ✔ | 76.2x50.8x23.6 for 5V-14V models, 76.2x50.8x22.5 for 15V-48V modelsTop Diagram i | |
| GTM43007-AWWVV-F (D3 without standoffs) | Open Frame/Internal | 100-240V~, 50-60 Hz | 60 | 5-48 V | N/A | N/A | ✔ | ✔ | ✔ | 76.2x50.8x23.6 for 5V-14V models, 76.2x50.8x22.5 for 15V-48V modelsTop Diagram i | |
| GTM43007-AWWVV-FW | Open Frame/Internal | 100-240V~, 50-60 Hz | 60 | 5-48 V | N/A | N/A | ✔ | ✔ | ✔ | 76.2x50.8x23.6 for 5V-14V models, 76.2x50.8x22.5 for 15V-48V models | |
| GTM43007-AWWVV-FW (D2 without standoffs) | Open Frame/Internal | 100-240V~, 50-60 Hz | 60 | 5-48 V | N/A | N/A | ✔ | ✔ | ✔ | 76.2x50.8x23.6 for 5V-14V models, 76.2x50.8x22.5 for 15V-48V models | |
| GTM43007-BWWVV-F | Open Frame/Internal | 100-240V~, 50-60 Hz | 60 | 5-48 V | N/A | N/A | ✔ | ✔ | ✔ | 101.6x50.8x23.6 for 5V-14V models, 101.6x50.8x22.5 for 15V-48V models | |
| GTM43007-BWWVV-FW | Open Frame/Internal | 100-240V~, 50-60Hz | 60 | 5-48 V | N/A | N/A | ✔ | ✔ | ✔ | 101.6x50.8x23.6 for 5V-14V models, 101.6x50.8x22.5 for 15V-48V models | |
| GTM43007-CWWVV-F | Open Frame/Internal | 100-240V~, 50-60 Hz | 60 | 5-48 V | N/A | N/A | ✔ | ✔ | ✔ | 127x76.2x23.6 | |
| GTM43007-CWWVV-FW | Open Frame/Internal | 100-240V~, 50-60 Hz | 60 | 5-48 V | N/A | N/A | ✔ | ✔ | ✔ | 127x76.2x23.6 | |
| GTM91099-60VV-P2 | Potted/Encapsulated in plastic housing | 100-240V~, 50-60 Hz | 60 | 5-48 V | V | IP68 | ✔ | ✔ | 144.81*57.11*39.0 | ||
| GTM91099-60VV-P3 | Potted/Encapsulated in plastic housing | 100-240V~, 50-60 Hz | 60 | 5-48 V | V | ✔ | ✔ | ||||
| GTM91099-60VV-P3(SR with NA Plug 3 pin) | Potted/Encapsulated in plastic housing | 100-240V~, 50-60 Hz | 60 | 5-48 V | V | ✔ | ✔ | ||||
| GTM91099-60VV-T2 | Wall Plug-in+Desktop Combination | 100-240V~, 50-60 Hz | 60 | 5-48 V | V | IP42 | ✔ | ✔ | 117.53*53.47*36.6 | ||
| GTM91099-60VV-T3A | Wall Plug-in+Desktop Combination | 100-240V~, 50-60 Hz | 60 | 5-48 V | V | IP52, IP54 Option available | ✔ | ✔ | ✔ | 117.53*53.47*36.6 | |
| GTM96600-60VV-P2(SR with NA Plug 2 pin) | Potted/Encapsulated in plastic housing | 100-240V~, 50-60 Hz | 60 | 5-48 V | USA DOE Level VI / Eco-design Directive 2009/125/EC, (EU) 2019/1782 | IP68 | ✔ | ✔ | 114.81*57.11*39.0 | ||
| GTM96600-60VV-P3(SR with fixed Plug 3 pin) | Potted/Encapsulated in plastic housing | 100-240V~, 50-60 Hz | 60 | 0 V | USA DOE Level VI / Eco-design Directive 2009/125/EC, (EU) 2019/1782 | IP68 | ✔ | ✔ | ✔ | 114.81*57.11*39.0 | |
| GTM96600-60VV.V-R2 | Wall Plug-in+Desktop Combination | 100-240V~, 50-60 Hz | 60 | 5-54 V | USA DOE Level VI / Eco-design Directive 2009/125/EC, (EU) 2019/1782 | IP40, IP54 Option available | ✔ | ✔ | ✔ | 117.53*53.47*36.6 | |
| GTM96600-60VV.V-R3A | Wall Plug-in+Desktop Combination | 100-240V~, 50-60 Hz | 60 | 5-54 V | USA DOE Level VI / Eco-design Directive 2009/125/EC, (EU) 2019/1782 | IP40, IP54 Option available | ✔ | ✔ | ✔ | 117.53*53.47*36.6 | |
| GTM96600-60VV.V-T2 | Desktop/External | 100-240V~, 50-60Hz | 60 | 5-54 V | USA DOE Level VI / Eco-design Directive 2009/125/EC, (EU) 2019/1782 | IP41 | ✔ | ✔ | ✔ | 117.5*53.5*34.2 | |
| GTM96600-60VV.V-T2A | Desktop/External | 100-240V~, 50-60Hz | 60 | 5-54 V | USA DOE Level VI / Eco-design Directive 2009/125/EC, (EU) 2019/1782 | IP41 | ✔ | ✔ | ✔ | 117.5*53.5*34.2 | |
| GTM96600-60VV.V-T3 | Desktop/External | 100-240V~, 50-60Hz | 60 | 5-54 V | USA DOE Level VI / Eco-design Directive 2009/125/EC, (EU) 2019/1782 | IP41 | ✔ | ✔ | ✔ | 117.5*53.5*34.2 | |
| GTM96600-60VV.V-T3A | Desktop/External | 100-240V~, 50-60Hz | 60 | 5-54 V | USA DOE Level VI / Eco-design Directive 2009/125/EC, (EU) 2019/1782 | IP41 | ✔ | ✔ | 117.5*53.5*34.2 | ||
| GTM96600-65VV-P2(SR with fixed Plug 2 pin) | Potted/Encapsulated in plastic housing | 100-240V~, 50-60 Hz | 60 | 0 V | USA DOE Level VI / Eco-design Directive 2009/125/EC, (EU) 2019/1782 | IP68 | ✔ | ✔ | ✔ | 114.81*57.11*39.0 | |
| GTM96605-G2-R2 | Wall Plug-in+Desktop Combination | 100-240V~, 50-60Hz | 60 | 0 V | USA DOE Level VI / Eco-design Directive 2009/125/EC, (EU) 2019/1782 | ✔ | ✔ | ✔ | |||
| GTM96605-G2-R3A | Wall Plug-in+Desktop Combination | 100-240V~, 50-60Hz | 60 | 0 V | USA DOE Level VI / Eco-design Directive 2009/125/EC, (EU) 2019/1782 | ✔ | ✔ | ✔ | 117.53*53.47*36.6 | ||
| GTM96605-G2-T2 | Desktop/External | 100-240V~, 50-60Hz | 60 | 0 V | USA DOE Level VI / Eco-design Directive 2009/125/EC, (EU) 2019/1782 | ✔ | ✔ | ✔ | |||
| GTM96605-G2-T3 | Desktop/External | 100-240V~, 50-60Hz | 60 | 0 V | USA DOE Level VI / Eco-design Directive 2009/125/EC, (EU) 2019/1782 | ✔ | ✔ | ✔ | |||
| GTM96600-65VV.V-P2 | Potted/Encapsulated in plastic housing | 100-240V~, 50-60Hz | 65 | 5-54 V | USA DOE Level VI / Eco-design Directive 2009/125/EC, (EU) 2019/1782 | IP68 | ✔ | ✔ | ✔ | 114.81*57.11*39.00 | |
| GTM96600-65VV.V-P3 | Potted/Encapsulated in plastic housing | 100-240V~, 50-60Hz | 65 | 5-54 V | USA DOE Level VI / Eco-design Directive 2009/125/EC, (EU) 2019/1782 | IP68 | ✔ | ✔ | 114.81*57.11*39.00 | ||
| GTM96600-65VV.V-R2 | Wall Plug-in+Desktop Combination | 100-240V~, 50-60 Hz | 65 | 12-48 V | USA DOE Level VI / Eco-design Directive 2009/125/EC, (EU) 2019/1782 | IP40, IP54 Option available | ✔ | ✔ | ✔ | 117.53*53.47*36.6 | |
| GTM96600-65VV.V-R3A | Wall Plug-in+Desktop Combination | 100-240V~, 50-60 Hz | 65 | 5-48 V | USA DOE Level VI / Eco-design Directive 2009/125/EC, (EU) 2019/1782 | IP40, IP54 Option available | ✔ | ✔ | ✔ | 117.53*53.47*36.6 | |
| GTM96600-65VV.V-T2 | Desktop/External | 100-240V~, 50-60Hz | 65 | 5-48 V | USA DOE Level VI / Eco-design Directive 2009/125/EC, (EU) 2019/1782 | IP41 | ✔ | ✔ | ✔ | 117.5*53.5*34.2 | |
| GTM96600-65VV.V-T2A | Desktop/External | 100-240V~, 50-60Hz | 65 | 5-48 V | USA DOE Level VI / Eco-design Directive 2009/125/EC, (EU) 2019/1782 | IP41 | ✔ | ✔ | ✔ | 117.5*53.5*34.2 | |
| GTM96600-65VV.V-T3 | Desktop/External | 100-240V~, 50-60Hz | 65 | 5-54 V | USA DOE Level VI / Eco-design Directive 2009/125/EC, (EU) 2019/1782 | IP41 | ✔ | ✔ | ✔ | 117.5*53.5*34.2 | |
| GTM96600-65VV.V-TP(SR with fixed Plug 2 pin) | Desktop/External | 100-240V~, 50-60Hz | 65 | 5-48 V | USA DOE Level VI / Eco-design Directive 2009/125/EC, (EU) 2019/1782 | IP42 | ✔ | ✔ | ✔ | 117.5*53.5*34.2 | |
| GTM41133-90VV-x.x-F | Open Frame/Internal | 100-240V~, 50-400Hz | 90 | 12-48 V | V | ✔ | ✔ | ✔ | 184*62*25 | ||
| GTM96900P90VV.V-T2 | Desktop/External | 100-240V~, 50-60 Hz | 90 | 12-54 V | USA DOE Level VI / Eco-design Directive 2009/125/EC, (EU) 2019/1782 | IP41 | ✔ | ✔ | 149.38 x 62.6 x 33.5 +/-1.0 | ||
| GTM96900P90VV.V-T3 | Desktop/External | 100-240V~, 50-60Hz | 90 | 12-54 V | USA DOE Level VI / Eco-design Directive 2009/125/EC, (EU) 2019/1782 | IP41 | ✔ | ✔ | ✔ | 149.3 x 62.8 x 34.0 +/-1.0 | |
| GTM96900P90VV.V-T3A | Desktop/External | 100-240V~, 50-60Hz | 90 | 12-54 V | USA DOE Level VI / Eco-design Directive 2009/125/EC, (EU) 2019/1782 | IP41 | ✔ | ✔ | ✔ | 149.38 x 62.6 x 33.5 +/-1.0 mm | |
| GTM96900P90VV.V-TP(SR with fixed Plug 2 pin) | Desktop/External | 100-240V~, 50-60 Hz | 90 | 12-24 V | USA DOE Level VI / Eco-design Directive 2009/125/EC, (EU) 2019/1782 | IP42 | ✔ | ✔ | 149.3 x 62.8 x 34.0 +/-1.0 | ||
| GTM96900P90VV.V-TP3(SR with fixed Plug 3 pin) | Desktop/External | 100-240V~, 50-60 Hz | 90 | 19 V | USA DOE Level VI / Eco-design Directive 2009/125/EC, (EU) 2019/1782 | IP42 | ✔ | ✔ | 149.3 x 62.8 x 34.0 +/-1.0 | ||
| GTD9X1002-WWWVV-TW | Desktop/External | 12-36V | 100 | 12-54 V | IP43 | ✔ | ✔ | ✔ | ✔ | 149.4 x 62.6 x 33.5 | |
| GTD9X1003-WWWVV-TW | Desktop/External | 12-36V | 100 | 12-54 V | IP43 | ✔ | ✔ | ✔ | ✔ | 149.4 x 62.6 x 33.5 | |
| GTM9X1001P-WWWVV-T2A | Desktop/External | 100-240V∿ @ 50-60Hz + 115V∿ @ 400Hz | 100 | 12-54 V | IP43 | ✔ | ✔ | ✔ | 149.4 x 62.6 x 34 | ||
| GTM961200P120VV.V-T2 | Desktop/External | 100-240V~, 50-60 Hz | 120 | 12-54 V | USA DOE Level VI / Eco-design Directive 2009/125/EC, (EU) 2019/1782 | IP41 | ✔ | ✔ | ✔ | 149.30*62.80*34.00 | |
| GTM961200P120VV.V-T3 | Desktop/External | 100-240V~, 50-60 Hz | 120 | 12-54 V | USA DOE Level VI / Eco-design Directive 2009/125/EC, (EU) 2019/1782 | IP41 | ✔ | ✔ | ✔ | 149.30*62.80*34.00 | |
| GTM961200P120VV.V-T3A | Desktop/External | 100-240V~, 50-60 Hz | 120 | 12-54 V | USA DOE Level VI / Eco-design Directive 2009/125/EC, (EU) 2019/1782 | IP41 | ✔ | ✔ | ✔ | 149.30*62.80*34.00 | |
| GTM961200P120VV.V-TP(SR with fixed Plug 2 pin) | Desktop/External | 100-240V~, 50-60 Hz | 120 | 42 V | USA DOE Level VI / Eco-design Directive 2009/125/EC, (EU) 2019/1782 | IP42 | ✔ | ✔ | 149.30*62.80*34.0 | ||
| GTM961600PWWWVV.V-T2 | Desktop/External | 100-240V~, 50-60 Hz | 160 | 12-36 V | USA DOE Level VI / Eco-design Directive 2009/125/EC, (EU) 2019/1782 | IP41 | ✔ | ✔ | ✔ | 176.5*74*40 | |
| GTM961600PWWWVV.V-T3 | Desktop/External | 100-240V~, 50-60 Hz | 160 | 12-54 V | USA DOE Level VI / Eco-design Directive 2009/125/EC, (EU) 2019/1782 | IP41 | ✔ | ✔ | ✔ | 176.5*74*40 | |
| GTM961600PWWWVV.V-T3A | Desktop/External | 100-240V~, 50-60 Hz | 160 | 12-36 V | USA DOE Level VI / Eco-design Directive 2009/125/EC, (EU) 2019/1782 | IP41 | ✔ | ✔ | ✔ | 176.5*74*40 | |
| GTM961800PWWWVV.V-T2 | Desktop/External | 100-240V~, 50-60 Hz | 180 | 12-54 V | USA DOE Level VI / Eco-design Directive 2009/125/EC, (EU) 2019/1782 | IP41 | ✔ | ✔ | ✔ | 176.5*74*40 | |
| GTM961800PWWWVV.V-T2A | Desktop/External | 100-240V~, 50-60 Hz | 180 | 12-54 V | USA DOE Level VI / Eco-design Directive 2009/125/EC, (EU) 2019/1782 | IP41 | ✔ | ✔ | ✔ | 176.5*74*40 | |
| GTM961800PWWWVV.V-T3 | Desktop/External | 100-240V~, 50-60Hz | 180 | 12-54 V | USA DOE Level VI / Eco-design Directive 2009/125/EC, (EU) 2019/1782 | IP41 | ✔ | ✔ | ✔ | 176.5*74*40 | |
| GTM961800PWWWVV.V-T3A | Desktop/External | 100-240V~, 50-60 Hz | 180 | 15-54 V | USA DOE Level VI / Eco-design Directive 2009/125/EC, (EU) 2019/1782 | IP41 | ✔ | ✔ | ✔ | 176.5*74*40 | |
| GTM962250PWWWVVX-XF | Open Frame/ L Frame/Cage/ Potted Internal | 100-240V~, 50-60 Hz | 225 | 12-54 V | USA DOE Level VI / Eco-design Directive 2009/125/EC, (EU) 2019/1782 | N/A | ✔ | ✔ | ✔ | 101.6 (L) x 50.8 (W) x 31.0 (H) | |
| GTM962251PWWWVVX-XF | Open Frame/Internal | 100-240V~, 50-60 Hz | 225 | 12-54 V | USA DOE Level VI / Eco-design Directive 2009/125/EC, (EU) 2019/1782 | na | ✔ | ✔ | ✔ | 103.5 (L) x 62.3 (W) x 40.0 (H) | |
| GTM962252PWWWVVX-XF | Open Frame/Internal | 100-240V~, 50-60 Hz | 225 | 12-54 V | USA DOE Level VI / Eco-design Directive 2009/125/EC, (EU) 2019/1782 | na | ✔ | ✔ | ✔ | 103.5 (L) x 62.3 (W) x 40.0 (H) | |
| GTM962253PWWWVV-P3 | Potted | 100-240V~, 50-60 Hz | 225 | 12-54 V | USA DOE Level VI / Eco-design Directive 2009/125/EC, (EU) 2019/1782 | IP68 | ✔ | ✔ | 181.0 (L) x 70.0(W) x 39.0 (H) | ||
| GTM962254PWWWVVX-XF | Open Frame/ L Frame/Cage/ Potted Internal | 100-240V~, 50-60 Hz | 225 | 24 V | USA DOE Level VI / Eco-design Directive 2009/125/EC, (EU) 2019/1782 | N/A | ✔ | ✔ | ✔ | 101.6 (L) x 50.8 (W) x 31.0 (H) | |
| GTM962500PWWWVV.V-CX | Open Frame/Internal | 100-240V~, 50-60 Hz | 250 | 15-36 V | USA DOE Level VI / Eco-design Directive 2009/125/EC, (EU) 2019/1782 | na | ✔ | ✔ | 203*63.5*41 | ||
| GTM962500PWWWVV.V-F | Open Frame/Internal | 100-240V~, 50-60 Hz | 250 | 12-54 V | USA DOE Level VI / Eco-design Directive 2009/125/EC, (EU) 2019/1782 | na | ✔ | ✔ | 203*63.5*41 | ||
| GTM962500PWWWVV.V-FL | Open Frame/Internal | 100-240V~, 50-60 Hz | 250 | 12-54 V | USA DOE Level VI / Eco-design Directive 2009/125/EC, (EU) 2019/1782 | na | ✔ | ✔ | 203*63.5*41 | ||
| GTM963500PWWWVVX-FW | Open Frame/ L Frame/Cage/ Potted Internal | 90-264Vac (47 – 63Hz) | 350 | 0 V | USA DOE Level VI / Eco-design Directive 2009/125/EC, (EU) 2019/1782 | N/A | ✔ | ✔ | ✔ | 114.3 (L) x 63.5 (W) x 30.5 (H) | |
| GTM965500P-BP-WWWVV-XX | Open Frame/Internal | 100-240V~, 50-60 Hz | 550 | 12-54 V | USA DOE Level VI / Eco-design Directive 2009/125/EC, (EU) 2019/1782 | N/A | ✔ | ✔ | ✔ | 127.0 (L) x 76.2 (W) x 34.3 (H) | |
| GTM965500P-WWWVV-XX | Open Frame/Internal | 100-240V~, 50-60 Hz | 550 | 12-54 V | N/A | ✔ | ✔ | ✔ | 127.0 (L) x 76.2 (W) x 34.3 (H) |